How does a coin know to land heads half of the time?
Category: Physics Published: January 24, 2025
By: Christopher S. Baird, author of The Top 50 Science Questions with Surprising Answers and Associate Professor of Physics at West Texas A&M University

Christopher S. Baird.
A coin doesn't know anything. A fair coin is just as likely to land heads as to land tails, for an individual coin toss. Even if you have already tossed a coin twenty times and the result was twenty heads in a row, the next toss is still equally likely to be heads or to be tails. This is because a coin does not have a memory. Therefore, the outcomes of the previous tosses have no effect on the next toss. You may then wonder, "If I got twenty heads in a row, don't the next twenty tosses have to all end up tails in order to get the required result of 50% heads? How does the coin accomplish this without knowing what it's supposed to do and without remembering what it already did? Also, if the next twenty tosses aren't tails, how in the world do we end up with the final outcome being 50% tails, as required? When I actually do this in real life, I usually get the final outcome to be 50% heads, or something close to that!"
The truth is that the next twenty tosses do not have to be all heads. In fact, the next twenty tosses do not have to be anything. It's entirely possible that the next twenty tosses are also all heads, giving a total of forty heads in a row. Furthermore, it's entirely possible to flip a coin a hundred times and get all heads. This is extremely improbable, but it's not impossible. So what makes the final result of 100% heads so improbable if the coin has no memory? The key here is that if you are talking about individual final outcomes, then getting all heads is no more improbable than any other individual final outcome of a hundred coin tosses. In fact, because the coin has no memory, every individual final outcome is exactly as likely as any other.
For instance, if you flip a coin six times, then the final outcome being, "heads, heads, heads, heads, heads, heads," is exactly as probable as the final outcome being, "heads, tails, heads, tails, heads, tails," in that order. You might say, "But when I flip a coin six times in real life, I usually get three heads, or something close to that." That's true, but what you are saying is different from what I am saying. I'm not saying that getting three heads for six coin flips is just as probable as getting all heads. I'm saying that the exact result, "heads, tails, heads, tails, heads, tails," in that order, is just as likely as getting all heads. The order matters!
If I got three heads after tossing a coin six times, but in the order, "heads, heads, heads, tails, tails, tails," then that's actually a different individual final outcome from getting, "heads, tails, heads, tails, heads, tails," because the order is different. However, people usually don't care about the order in coin tosses, meaning that only the total number of heads matters. Considering only unordered final outcomes is what makes different probabilities come into play. If all of the order-specific final outcomes are equally likely, then the one particular unordered final outcome that is associated with the highest number of order-specific final outcomes will be the most likely to occur. In short, there's only one way to get all heads, but there's many different ways to get 50% heads. Let's go through some actual numbers to make this more concrete.
One Coin Flip
Consider a single coin flip in isolation. Either result, heads or tails, is equally likely. This means that we have a 1 in 2 chance of getting a heads and a 1 in 2 chance of getting a tails. There's nothing surprising here, so let's move on.
Two Coin Flips
Now consider two coin flips in a row. The individual final outcomes that are possible are shown below. Each of these individual final outcomes is equally likely because the coin has no memory. With four possible final outcomes, each outcome has a 1 in 4 chance.
- Heads and then Heads: 1 in 4 chance
- Heads and then Tails: 1 in 4 chance
- Tails and then Heads: 1 in 4 chance
- Tails and then Tails: 1 in 4 chance
If we don't care about the order—we just care about the total number of heads out of all of the coin flips—we should group together the individual final outcomes that lead to the same number of heads. For two coin flips in a row, the results are:
- All Heads: one way to get this result, giving a 1 in 4 chance
- Half Are Heads: two ways to get this result, giving a 2 in 4 chance
- No Heads: one way to get this result, giving a 1 in 4 chance
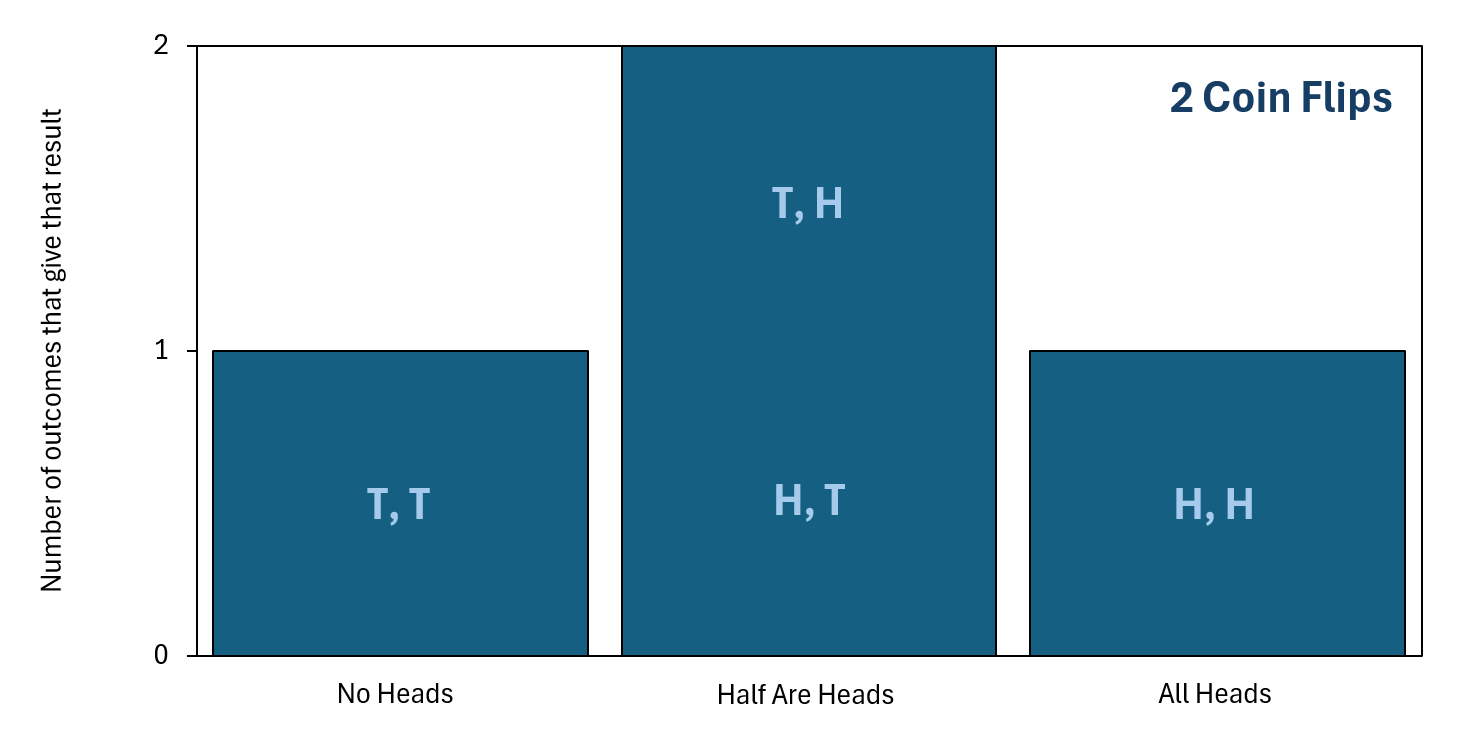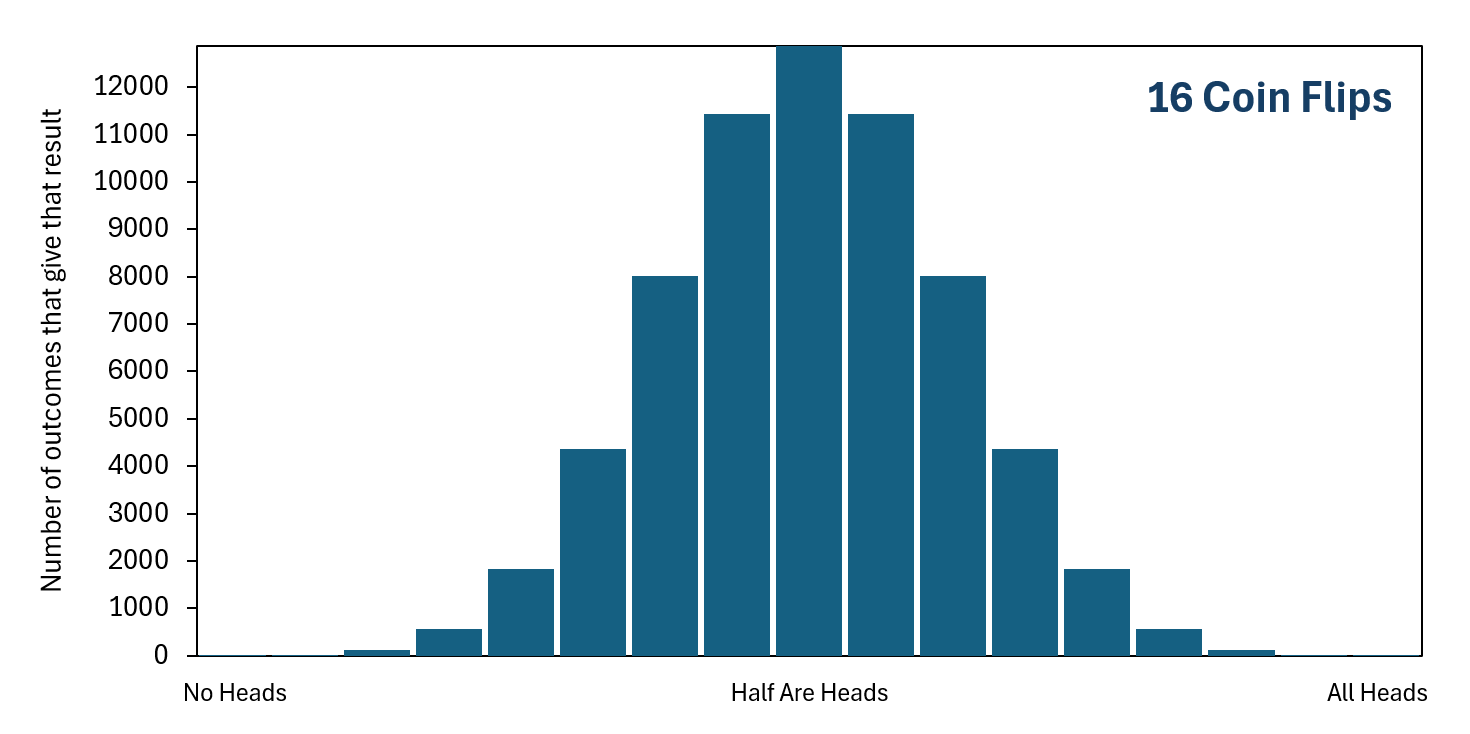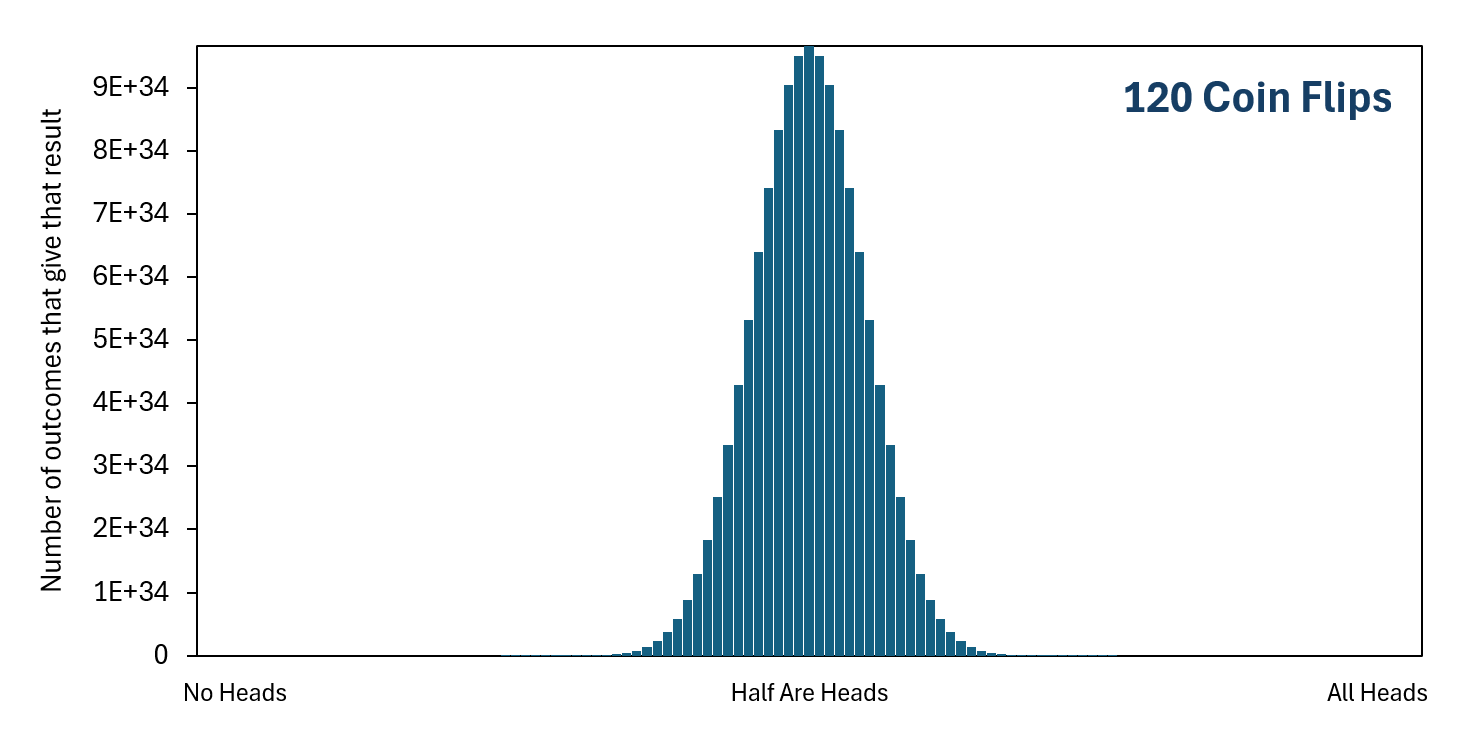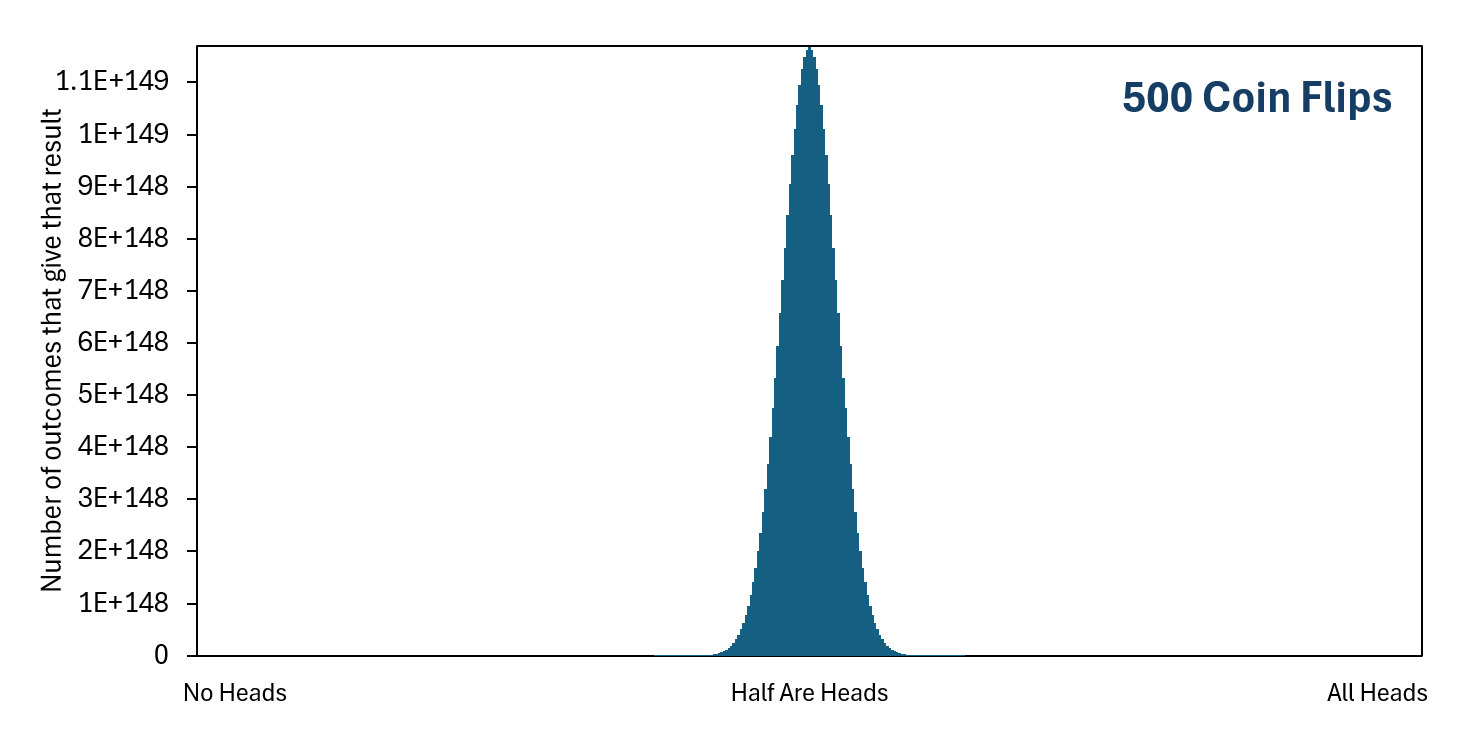
As you can see, half of the flips being heads (and the other half tails) is already the mostly likely unordered final outcome. This is because there are two different individual final outcomes that give the same unordered final outcome of half the flips being heads. To organize this information better, we can plot it as a bar chart, which is shown below. In this chart, "H" represents a flipped coin giving heads and "T" represents a flipped coin giving tails. The statement "T, H" represents getting a tails for the first coin flip and then a heads for the second coin flip, and other statements like this follow the same notation.
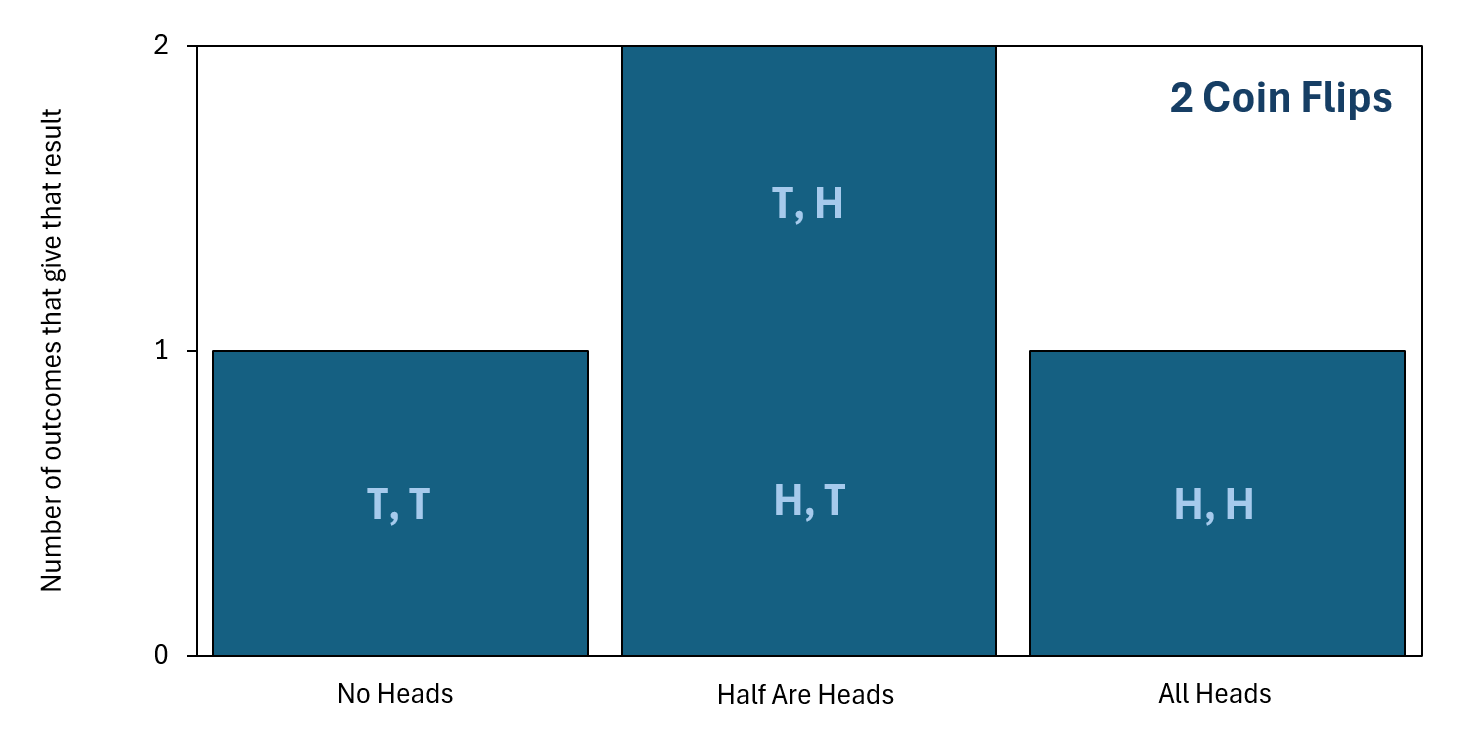
so that order does not matter. Click the image to enlarge it.
Public Domain Image, source: Christopher S. Baird.
As the bar chart makes clear, even with just two coin flips, getting the final outcome to be "Half Are Heads" is the most likely result. This is simply because there are two ways to get "Half Are Heads" but only one way to get "All Heads" and only one way to get "No Heads." With that said, this doesn't guarantee that you'll always get "Half Are Heads." In fact, for just two coin flips, getting "All Heads" is quite likely—it has a probability of 25%, to be exact.
Three Coin Flips
Next consider three coin flips in a row. The individual final outcomes that are possible are shown below. Each of these individual final outcomes is equally likely because the coin has no memory. With eight possible final outcomes, each individual final outcome has a 1 in 8 chance.
- Heads, Heads, Heads: 1 in 8 chance
- Tails, Heads, Heads: 1 in 8 chance
- Heads, Tails, Heads: 1 in 8 chance
- Heads, Heads, Tails: 1 in 8 chance
- Tails, Tails, Heads: 1 in 8 chance
- Tails, Heads, Tails: 1 in 8 chance
- Heads, Tails, Tails: 1 in 8 chance
- Tails, Tails, Tails: 1 in 8 chance
If we don't care about the order, we should again group together the individual final outcomes that lead to the same number of heads. For three coin flips in a row, the results are:
- All Heads: one way to get this result, giving a 1 in 8 chance
- Two Thirds Are Heads: three ways to get this result, giving a 3 in 8 chance
- One Third Are Heads: three ways to get this result, giving a 3 in 8 chance
- No Heads: one way to get this result, giving a 1 in 8 chance
We can again plot this information in a bar chart, which is shown below. The statement "T, T, H" represents getting a tails for the first coin flip, and then a tails for the second coin flip, and then a heads for the third coin flip. Similar statements follow the same notation.
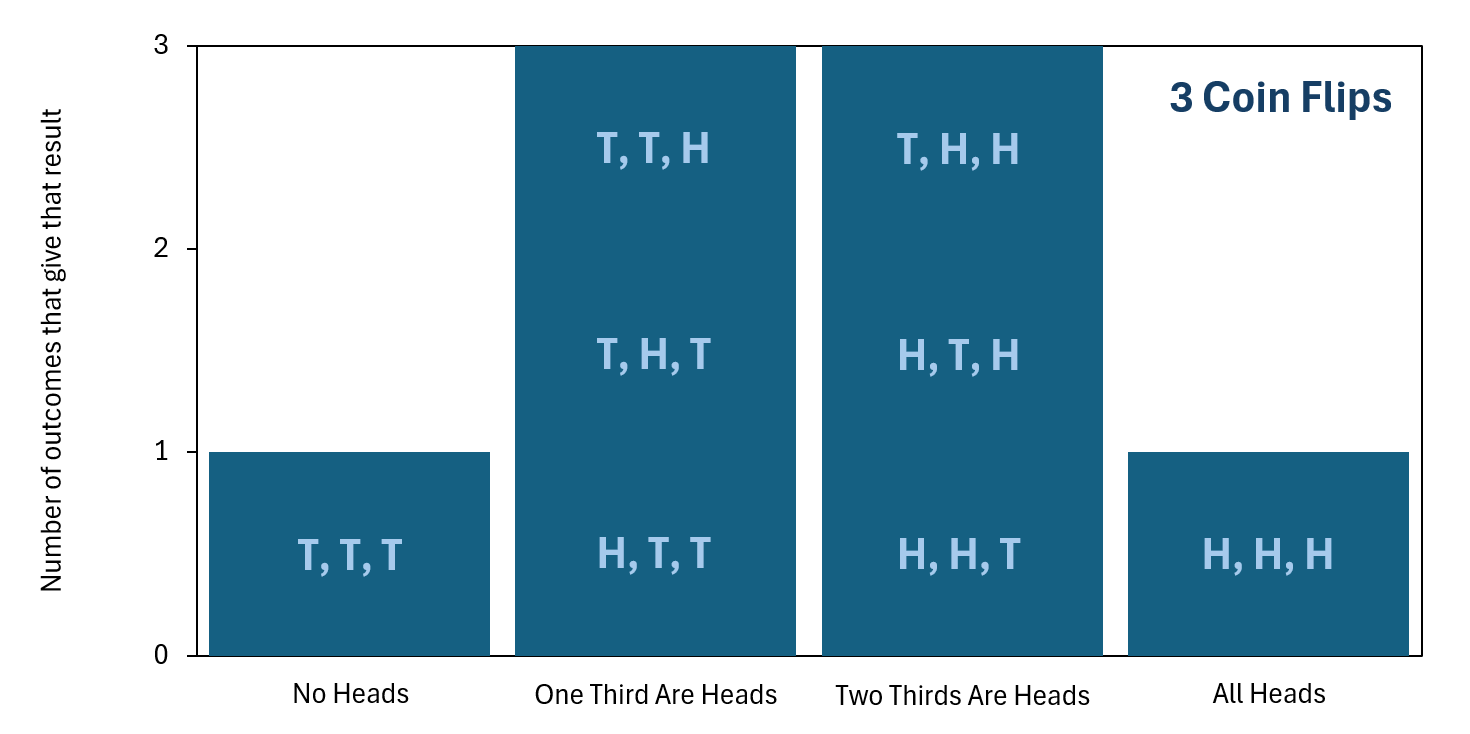
so that order does not matter. Click the image to enlarge it.
Public Domain Image, source: Christopher S. Baird.
Four Coin Flips
If we do the same type of analysis for four coin flips, then after having grouped together individual final outcomes that give the same number of heads, the results are:
- All Heads: one way to get this result, giving a 1 in 16 chance
- Three Fourths Are Heads: four ways to get this result, giving a 4 in 16 chance
- Half Are Heads: six ways to get this result, giving a 6 in 16 chance
- One Fourth Are Heads: four ways to get this result, giving a 4 in 16 chance
- No Heads: one way to get this result, giving a 1 in 16 chance
We then plot this information in a bar chart, which is shown below. As you can see, the outcome "Half Are Heads" is now even more probable than the outcome "No Heads" because there are so many different ways to get the outcome "Half Are Heads."
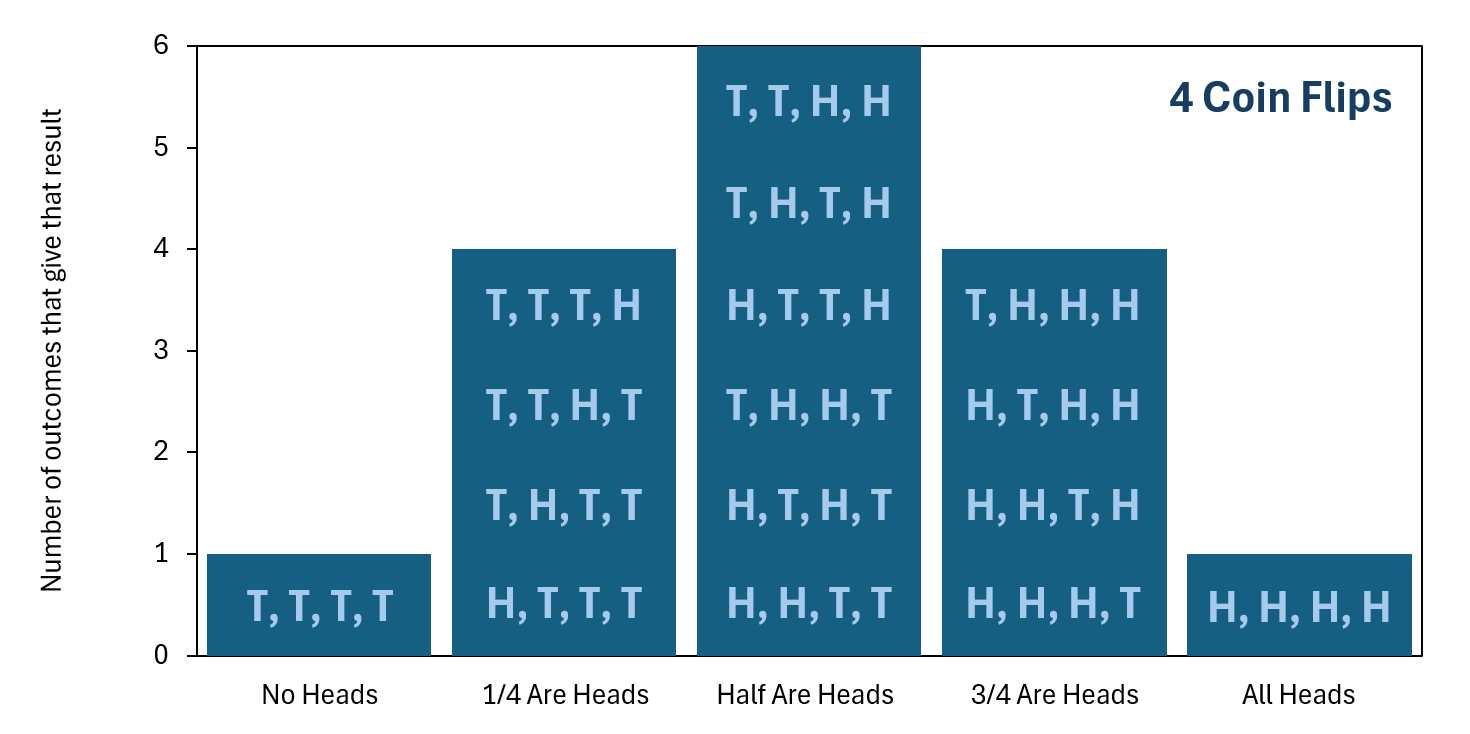
so that order does not matter. Click the image to enlarge it.
Public Domain Image, source: Christopher S. Baird.
Many Coin Flips
As we include more coin flips in our set of flips, getting "All Heads" becomes less probable very quickly because there are increasingly more ways to get one of the other unordered final outcomes, but still just one way to get "All Heads." In contrast, getting "Half Are Heads" becomes increasingly more likely than most of the other unordered final outcomes because there are increasingly more ways to get this result. With that said, getting exactly "Half Are Heads" becomes less likely overall as the number of coin flips increases, simply because there becomes more possible unordered final outcomes. But getting close to "Half Are Heads" becomes more likely overall as the number of coin flips increases. If you actually flip a coin a hundred times in real life, and then you do this over and over again, you will find it unlikely that you get exactly fifty heads, but likely that you will get something somewhat close to fifty heads.
If we do the exact same thing—list out all possible final outcomes and then group them according to the number of heads—for a set of five coin flips, and then a set of six coin flips, and then for a set of seven coin flips, and so forth, then we can plot each set and see how the pattern progresses. Rather than display dozens of separate images, I have stitched together the images into the animation shown below. Make sure you watch the animation below all the way until it reaches the set of 500 coin flips.
As you can see, the higher the number of coin flips there are in the set, the lower the probability of getting "All Heads" or of getting "No Heads" or of getting anything close to these. At the same time, the probability increases that you will get close to "Half Are Heads."
You may worry that this mathematical analysis won't hold up if you do real coin flips in the real world. To show you real-world results, I just flipped a coin a hundred times and got 54 heads. As expected, its not exactly 50 out of 100, but it's somewhat close. I just did it again repeatedly and I got 63 out of 100, then 51 out of 100, then 55 out of 100, then 45 out of 100, and then 39 out of 100, and then 52 out of a hundred. As you can see, getting exactly "Half Are Heads" (getting 50 heads out of 100 coin flips) did not happen, but getting close to "Half Are Heads" happened frequently. At the same time, getting "All Heads"or even close to this never happened and getting "No Heads" or even close to this never happened.

